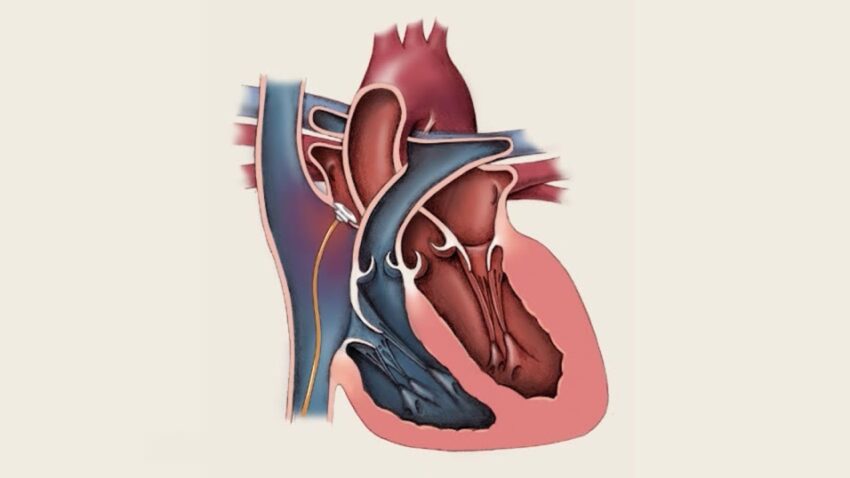
A patent foramen ovale (PFO) is a hole in the heart that did not close as expected after birth. Normally, prior to birth, an opening called the foramen ovale allows blood to bypass the lungs in utero. After birth, as an infant takes its first breath, pressure changes in the heart chambers cause the foramen ovale to close. In approximately 25 percent of the population, however, the foramen ovale fails to seal completely, leaving a PFO.
Most people who have a PFO do not experience any symptoms. In some cases, though, a Patent Foramen Ovale (PFO) Closure Devices allows blood clots to pass from the right side of the heart to the left side and travel to the brain, which can increase the risk of stroke. Studies have found that up to one-quarter of all strokes may be due to a PFO.
Detecting a Patent Foramen Ovale (PFO) Closure Devices
There are a few different tests that can diagnose a PFO. A bubble contrast echocardiogram uses an ultrasound to detect bubbles in the left side of the heart after they have been injected into a vein in the arm. If bubbles are seen, it indicates the presence of a PFO.
A transesophageal echocardiogram (TEE) provides clearer images of the heart than a standard echocardiogram. During a TEE, a probe is inserted into the esophagus, behind the heart. This allows ultrasound images to be taken from behind the heart and better visualize any PFO.
Magnetic resonance imaging (MRI) or computed tomography (CT) scans with contrast can also potentially spot a PFO, but echo tests are usually the preferred initial choice due to their low risk and good diagnostic accuracy.
PFO Closure Device Options
For patients at higher risk of recurrent stroke who have a Patent Foramen Ovale Closure Devices, closing the opening non-surgically with a PFO closure device may help reduce that risk. These minimally invasive procedures are typically performed in a hospital catheterization lab under local anesthesia. Several different types of PFO closure devices have been developed for this purpose:
– Amplatzer PFO Occluder: Made of self-expanding nitinol wire mesh framed by polyester fabric, this was one of the first PFO closure devices approved. It is shaped like a mushroom when deployed.
– Helex Septal Occluder: Another nitinol wire mesh device in a double-disk design. The two round disks are connected by a short central waist.
– CardioSEAL Septal Occlusion System: Unique “sandwich” design with polyester fabric disks inside a Nitinol wire mesh frame to block the PFO channel.
– Gore Cardioform Septal Occluder: Newer option made of ePTFE-coated nitinol frames held together with sutures, forming a “plug” shape.
How a PFO Closure Procedure Works
During a PFO closure procedure, patients are awake but sedated as the interventional cardiologist accesses the femoral or jugular vein in the groin or neck. Special catheters and guidewires are used to navigate through the vein and into the heart chambers under X-ray guidance.
Echocardiography helps the doctor visualize the PFO channel. Then the collapsed closure device is guided through a catheter and deployed straddling the PFO opening. Several devices may be used to ensure complete closure.
After deployment, another echocardiogram confirms the device is in the proper position, fully covering the PFO channel. The catheter is then removed and pressure is applied to the wound site to prevent bleeding. Most patients can go home the same day.
Outcomes of Patent Foramen Ovale Closure
Numerous clinical trials and studies have shown PFO closure to significantly reduce the risk of recurrent stroke compared to medical management alone in suitable patients. The largest randomized control trial to date, RESPECT, found a nearly 70% reduction in recurrent stroke or transient ischemic attack (TIA) over 5 years in patients who underwent PFO closure versus anti-clotting medications.
Procedure success rates for closing the defect are high, generally over 95%. Major complications are rare, occurring in less than 1% of cases, but can include device embolization, cardiac perforation or tamponade. Most common side effects include bruising, nausea or headache and resolve quickly. Long-term data also indicates closure devices have a very low rate of complications over time.
Overall, PFO closure has become an accepted treatment when stroke is felt to be caused by a PFO in adults under 60, who have had an otherwise unexplained ischemic stroke. It provides effective reduction in risk for these patients with minimal side effects compared to open heart surgery.
patent foramen ovale is a common congenital heart abnormality usually not requiring treatment. But for patients with certain risk factors like a history of cryptogenic stroke, PFO closure can successfully reduce future stroke risk by sealing the opening in a minimally invasive transcatheter procedure. The variety of FDA-approved devices available allows interventional cardiologists to choose the best closure option based on each patient’s specific PFO anatomy. When performed by an experienced operator, PFO closure has proven highly effective at preventing recurrent neurological events safely long-term.
About Author - Ravina Pandya
Ravina Pandya,a content writer, has a strong foothold in the market research industry. She specializes in writing well-researched articles from different industries, including food and beverages, information and technology, healthcare, chemicals and materials, etc. With an MBA in E-commerce, she has expertise in SEO-optimized content that resonates with industry professionals. LinkedIn Profile